|
The formal charge is a theoretical charge and thus does not indicate any real charge separation present in the molecule. Thus below are the steps to draw the Lewis structure.ĭetermine the number of valence electrons present in the molecule. How to draw Lewis Structure: Steps with Exampleįormal Charge and its Mathematical formulaĬalculation of the Formal Charge for SO 4 -2īelow are the Steps to Find the Lewis Dot Structure.ĭrawing the Lewis structure is very important as it is then only when one can calculate the formal charge correctly. There are the following topics covered in this article: Learning to make proper Lewis dot structures can help in solving the problem that most of the students have. It also helps in predicting the geometry of the molecule. 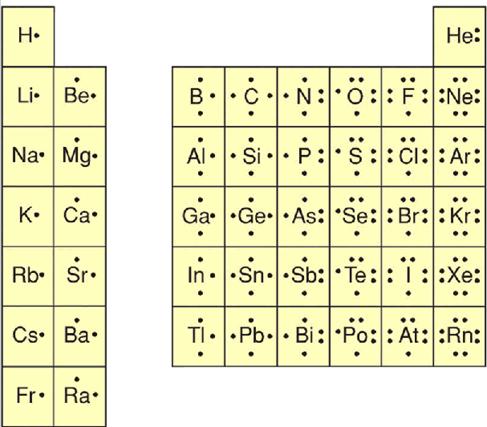 
The major reason why learning Lewis dot structure is important is that it helps in predicting the number and type of bonds which can be formed around an atom. Lewis structure is basically a graphic representation of the electron distribution around an atom.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
